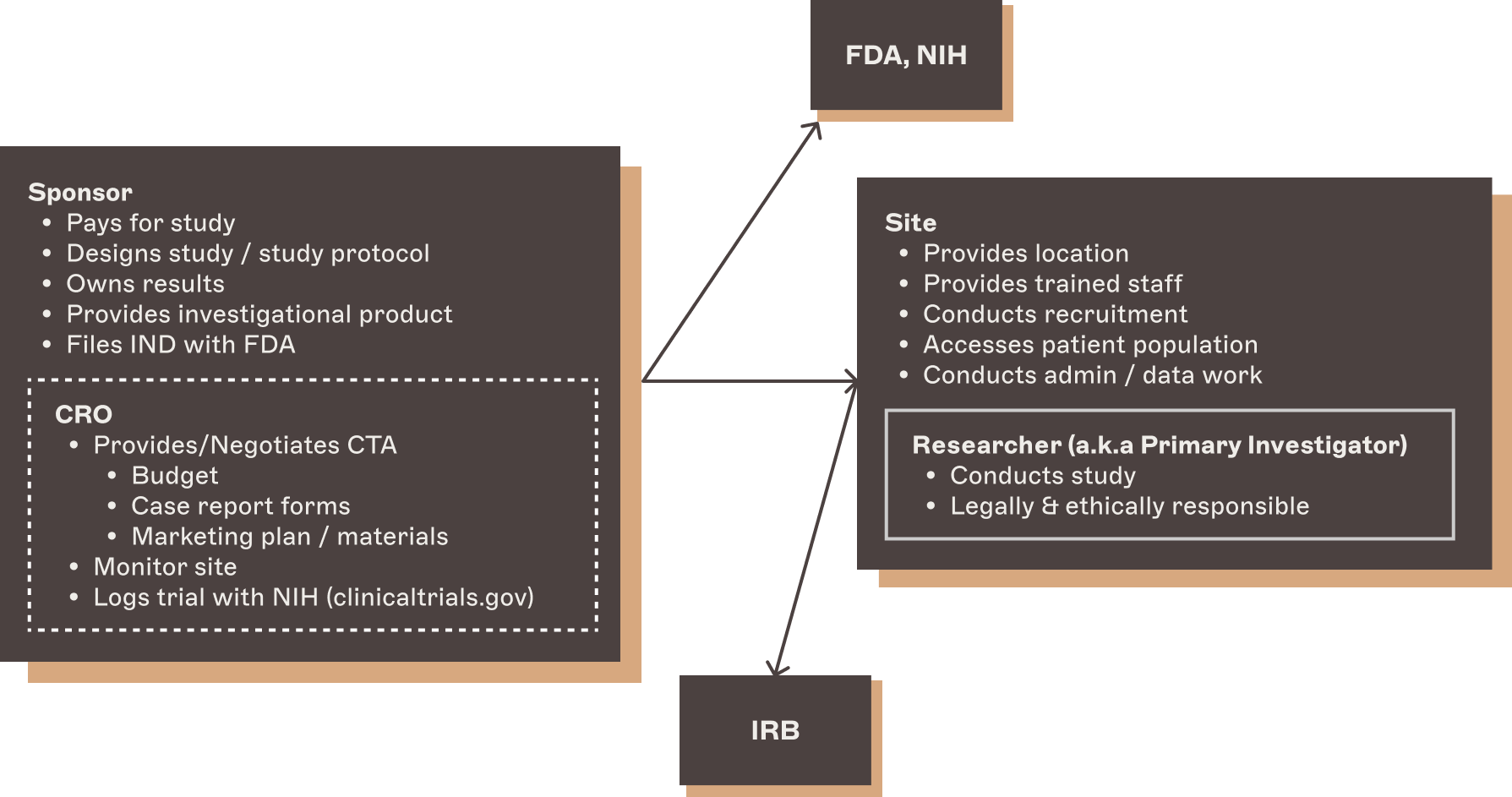
The basic parties involved in a clinical trial are the sponsor and the site.
The sponsor is the group that wants to conduct the research. For drug trials, the sponsor is usually the pharmaceutical company that makes the investigational drug. They design the study, pay for it, and ultimately own the results. The sponsor is the group that files the IND with FDA, which outlines the entire clinical research program across all phases and kicks off the whole process.
In many cases the sponsor will subcontract out a lot of the trial administration, like finding and liaising with sites, to a contract research organization (CRO). A CRO is a third-party partner that takes over most of the work to stand up and oversee each study—they manage the relationships with sites and investigators, negotiate clinical trial agreements, and often take over quality control, quality assurance, and monitoring duties.
The sponsor owns the interface with FDA and NIH, with the CRO usually taking point on keeping NIH and clinicaltrials.gov up to date. Even when working with a CRO, the sponsor remains responsible for the trial as a whole.
The site is the entity that enters into an agreement with the sponsor to actually do the research. "Site" is a slightly misleading name—a site can be a multilocation organization, so for example, an AIC contracting with a sponsor or CRO would be one site even if their research spans multiple physical locations. The site is responsible for all the actual activity that goes into the trial, like recruiting and seeing patients, collecting and reporting data, and administering the investigational drug.
One very important thing to understand is that patient recruitment is usually the responsibility of the site. We’ll cover this topic in substantial depth later in the guide.
At the site, the person who is responsible for the study is the researcher (or principal investigator, or PI). This is usually the site’s lead clinician. The researcher is the person who is ultimately personally responsible for the study, and is both legally and ethically responsible for the patients' wellbeing. They are usually, but not always, an employee of the site.
The last relevant party to mention is the Institutional Review Board (IRB). All studies have to be conducted under the watch of an IRB, which approves study protocols and must be kept up to date on key events. One of the factors leading to the pharma industry’s growing preference for placing studies with independent sites or site networks is their ability to work with a “non-local” or central IRB, which usually means a for-hire external group, who are much more nimble and less restrictive than local IRBs at research institutions. In practice, the main providers of central IRB services are WCG, Advarra, and Brany.